Top 7 Things to Know About Enbrel
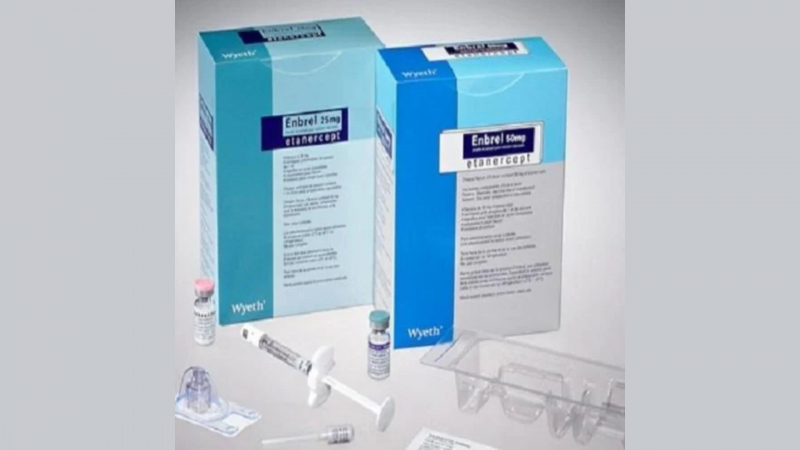
Enbrel is prepared as a powder for solution for injection, with the main ingredient being Etanercept. It is used to relieve the symptoms of advanced arthritis ... read more...in people with psoriatic arthritis. Check out the following useful information about Enbrel.
-
Enbrel is a brand name (trade name) for etanercept, which is used to treat inflammatory conditions like psoriasis and rheumatoid arthritis.
Enbrel works by impersonating the p75 TNF receptor. TNF (tumor necrosis factor) is a cytokine that occurs naturally (cytokines are small proteins that are involved in interactions and communications between cells). Cytokines and other proteins are involved in the development of many inflammatory conditions, including psoriasis and rheumatoid arthritis, as well as the subsequent joint destruction. TNF levels are elevated in the tissues and bodily fluids of people suffering from these conditions. TNF binds to one of two cell receptors known as p55 TNF receptors or p75 TNF receptors. TNF-alpha and TNF-beta bind to Enbrel rather than the cell surface TNF receptors because p75 serves as a dummy TNF receptor. TNF becomes inactive as a result.
Enbrel is a member of the group of drugs known as TNF inhibitors.

The Atlantic 
MIMS -
Licensed to treat moderate to severe polyarticular juvenile idiopathic arthritis (JIA) in children aged 2 and older and adults with moderate to severe active rheumatoid arthritis, either alone or in combination with methotrexate.
Adults and kids who are at least 4 years old and who qualify for systemic therapy or phototherapy can use it to treat moderate to severe plaque psoriasis. Adults with psoriatic arthritis can use it with or without methotrexate to lessen signs and symptoms, stop structural progression, and enhance physical function.
It has been approved for easing the symptoms and indications of active ankylosing spondylitis.
Enbrel contains a fragment of human immunoglobulin G1, which gives it a more profound and long-lasting effect than naturally occurring soluble TNF receptors.
It can be administered by a doctor or nurse, or people can be taught to do it themselves.
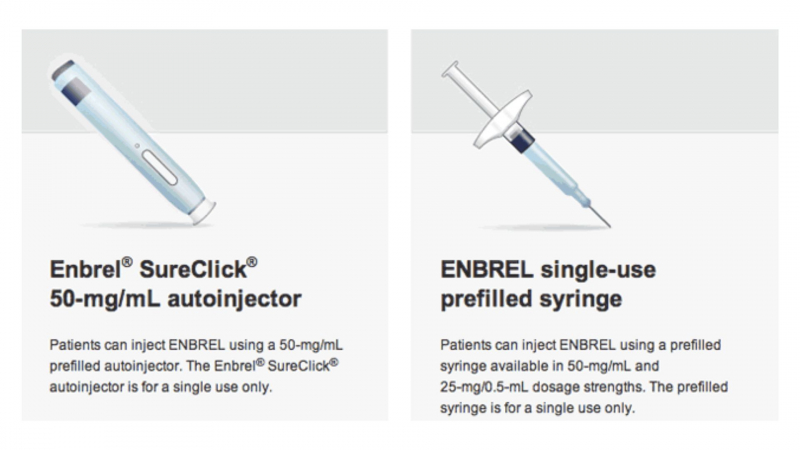
Enbrel mini single-dose prefilled cartridges for use with the AutoTouch reusable autoinjector are available as a single-dose vial, a single-dose prefilled syringe, a single-dose prefilled SureClick autoinjector, and an Enbrel mini single-dose prefilled syringe. Administration is simple to grasp.
Adult rheumatoid arthritis, ankylosing spondylitis, and psoriatic arthritis: 50mg once weekly for three months, then once weekly thereafter; psoriasis: 50mg twice weekly for three months, then once weekly thereafter.
Less pain, stiffness, and edema are among the effects, along with improvements in mobility and inflammatory laboratory indicators.
The Atlantic 
Arthritis Rheumatism -
You are more likely to have the following side effects if you are between the ages of 18 and 60, do not take any other medications, or have any other medical conditions:
- The most common side effects reported are injection site reactions (such as redness, pain, and swelling at the injection site) and upper respiratory tract infections.
- According to post-marketing reports, Enbrel increases a person's risk of infection by about 20%. Invasive fungal infections, such as histoplasmosis, coccidioidomycosis, candidiasis, aspergillosis, blastomycosis, and pneumocystosis; bacterial infections; viral infections; and other infections, such as legionella or listeria, have been reported. People with a history of chronic or recurring infection may be ineligible.
- Before starting Enbrel, all patients should be tested for latent tuberculosis and, if positive, treatment should be started.
- Before beginning Enbrel treatment, ensure that all patients have received all of the appropriate immunizations for their age, as recommended by national guidelines. It is critical that people who have been given Enbrel do not receive live vaccines.
- Because it is such an unstable molecule, it is unable to be administered orally (as are most other biologics). This is why it must be administered via injection.
- Some people, including those with a history of hepatitis B, heart failure, or nervous system problems, may be ineligible.
- Enbrel is linked to an increased risk of lymphoma and other cancers.
- Must be kept cold (between 2°C and 8°C; 36°F and 46°F). To protect against light, store in the original carton. Can be maintained at room temperature for up to 14 days at a maximum of 77°F (25°C). Avoid shaking, heating, freezing, and exposing to direct sunlight. Don't put Enbrel back in the fridge after storing it at room temperature, and throw it away if you don't use it within 14 days.
- It is unknown how Enbrel affects a developing baby, but your doctor may recommend it if the benefits outweigh the risks. If you become pregnant while taking Enbrel, notify your doctor right away. It is unknown whether previous Enbrel administration affects live vaccinations given to newborns shortly after birth. Enbrel is found in low concentrations in human milk and is only slightly absorbed by a breastfed infant. There are no data on its effects.
- There is no generic version of Enbrel available.
In general, those who are older or younger, have specific medical conditions (including liver or kidney issues, heart disease, diabetes, or seizure disorders), use other medications, or are seniors or young children are more likely to experience a greater range of adverse effects.

NiceRx 
Cleveland Clinic -
Before beginning Enbrel treatment, make sure you have read the Enbrel Medication Guide. Before treatment, ask any questions you may have.
You can administer Enbrel yourself if your healthcare provider has shown you how to do so and you are comfortable doing so. Remove Enbrel from the refrigerator and allow it to come to room temperature for 30 minutes before using it. This will make the injection more bearable. Warming it up in the microwave or with hot water will destroy the medicine. The Enbrel injection should not be shaken.
Enbrel should only be injected into the outer area of your upper arm (if somebody else is administering it to you), your thigh, or your stomach (apart from the 2 inch [5 cm] area surrounding your belly button) . Use soap and water to wash your hands.
Through the viewing window on your Enbrel autoinjector, check the injection status. The interior liquid should appear transparent and colorless. The medication may contain a few tiny white flecks. If the medication is foggy, discolored, or contains big particles, do not use it. Additionally, make sure to look at the autoinjector's expiration date, which is printed on the side.
Pick a place to inject yourself, then clean it with an alcohol wipe. Dry it out. Use caution when applying to skin that is rashes, bruised, broken, covered in psoriatic lesions, or discolored. Straighten up the Enbrel autoinjector cap and remove it. The drug may dry out if the cap is left off for longer than five minutes. Stretch the skin out with your fingers or pinch it up to create a solid surface at the injection site. Put the autoinjector at a 90-degree angle (straight up and down) on the injection site of your choice. Hold down the Enbrel autoinjector while applying firm pressure. Press the purple start button once you're ready to inject. The needle will be inserted and the injection will begin. A clicking sound will be heard. When the injection is complete, hold the autoinjector firmly down until the clicking sound stops and the viewing window changes from clear to yellow. You might hear another click. If the window does not change color, contact your doctor. Put your autoinjector and cap in an approved sharps container. Do not replace the autoinjector's cap. Each SureClick autoinjector is administered only once. After starting the injection, the status button on the AutoTouch reusable injector flashes green and then turns off.
People have reported stinging for up to 20 seconds after taking Enbrel. Allow the alcohol to evaporate off the injection site after cleaning it with an alcohol swab so that your skin is dry before administering Enbrel. Ice applied to the area before and after administration may also help reduce stinging. In general, injecting into the stomach is less painful than injecting into the thigh. To prevent scar tissue buildup, rotate injection sites. Allowing a family member or friend to administer the shots instead can help reduce the anticipation, making the procedure less painful.
If you encounter any of the following symptoms, which could mean you have an infection, let your doctor know right away: Coughing, diarrhea, fever, sweats, chills, muscular pains, shortness of breath, stomach pain, fatigue, warm, painful, or red skin, or sores on your body; or weight loss. Burning when you urinate or urinating more frequently than usual.
If you experience an allergic reaction after taking Enbrel, seek immediate medical attention (symptoms include anaphylaxis or urticaria). Latex is present in the needle cover of the prefilled syringe, the cover within the white cap of the SureClick, and the purple cap within the Enbrel mini cartridge.
If you have a cold, you can take Enbrel (etanercept), but you should notify your doctor because he or she may want to monitor your health while your cold symptoms are present, as you are at a higher risk of developing a more serious infection because you are taking Enbrel.
Until it is time to administer Enbrel, it should be kept in the refrigerator at a temperature of 36°F to 46°F (2°C to 8°C). Enbrel can be stored at room temperature for up to 14 days as long as it is shielded from light and is not heated. Do not re-refrigerate Enbrel after taking it out of the fridge for more than 15 minutes. After 14 days of being out of the fridge, Enbrel should be thrown away if not used. Always verify the expiration date and never provide expired Enbrel. Avoid freezing Enbrel.
Inform your doctor right away if you develop any new medical conditions (such as heart failure or autoimmune disorders) or if your current condition worsens.
It is unknown how Enbrel affects a developing baby, but your doctor may recommend it if the benefits outweigh the risks. If you become pregnant while taking Enbrel, notify your doctor right away.
Enbrel should not be stored in extreme temperatures, such as the glove box or trunk of a car.

Verywell Health 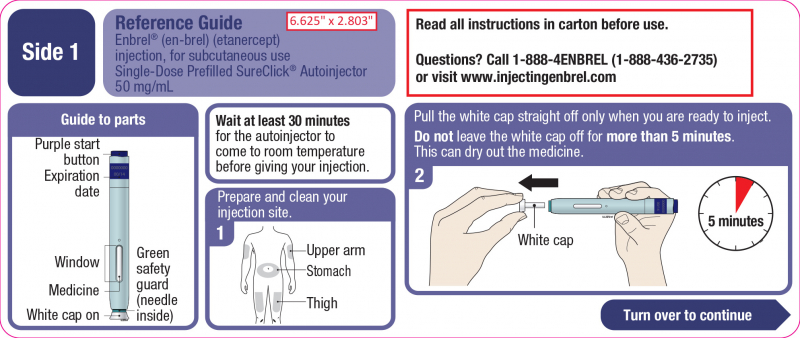
Enbrel® -
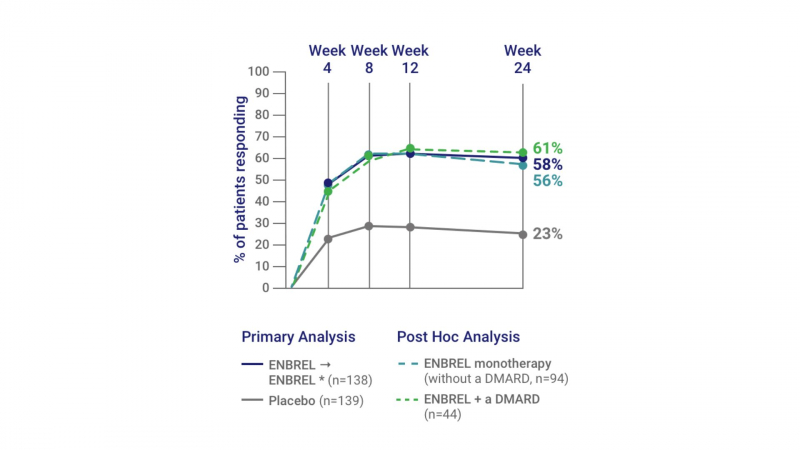
It begins to have an effect within one to two weeks, but the full effects may take 3 to 6 months to develop.
The American College of Rheumatology (ACR) response is a measurement used in trials looking at Enbrel's effectiveness. Here is how this is explained: ACR 50: A 50% reduction in the number of painful and swollen joints, as well as a 50% reduction in three of the five criteria listed below: erythrocyte sedimentation rate or C-reactive protein (CRP), patient global assessment, physician global assessment, functional ability measure (most commonly the Health Assessment Questionnaire (HAQ)), visual analog pain scale, and functional ability measure (functional ability measure). In other studies, by the third month, over 42% of participants had a confirmed clinical response (ACR 50) and 15% had a strong clinical response (ACR 70).
Additionally, studies have demonstrated that taking Enbrel along with methotrexate improves responsiveness.
Enbrel® (etanercept) 
Enbrel® (etanercept) -
Medicines that interact with Enbrel may reduce its effect, shorten its duration of action, increase side effects, or have no effect when combined. An interaction between two medications does not always necessitate the discontinuation of one of them; however, it can. Consult your doctor about how to handle drug interactions.
Enbrel interacts with over 387 medications, with the majority of these interactions being classified as major or moderate. Enbrel may interact with the following medications:
- benzodiazepines such as clonazepam or diazepam
- bleomycin
- capecitabine, carboplatin, cyclophosphamide
- cholesterol-lowering agents such as atorvastatin or fluvastatin
- cladribine
- heart medications such as amiodarone or nifedipine
- hormones such as ethinylestradiol or levonorgestrel
- hydroxyurea
- immunosuppressants such as azathioprine or cyclosporine
- interferon or peginterferon
- live vaccines such as measles, mumps, rubella, yellow fever, varicella, or zoster vaccines
- methotrexate
- opioids such as methadone
- other biologics, such as adalimumab, anakinra, abatacept, golimumab, or infliximab
- sulfasalazine
- steroids such as betamethasone and dexamethasone
- thalidomide
- theophylline
- warfarin.
People who have had significant varicella virus exposure should temporarily stop taking Enbrel and be considered for prophylactic treatment with varicella zoster immune globulin.
If a doctor has recommended antibiotics for a patient with a current bacterial illness, they may be used with this medication. While the patient is undergoing therapy for the illness, a doctor may decide to cease Enbrel, either temporarily or permanently, if the patient gets a major infection or sepsis.
Note that only commonly used drugs that may interact with Enbrel are included in this list, which is not exhaustive. For a comprehensive list of interactions, consult the Enbrel prescribing instructions.

NPR 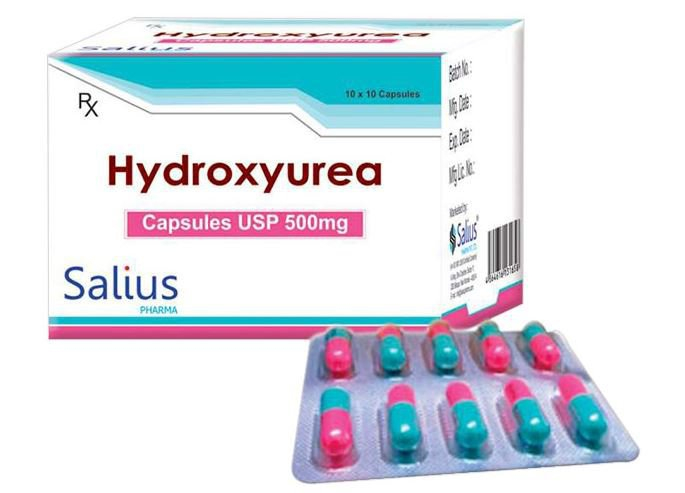
Vinmec