Top 8 Things to Know About Zubsolv
If you're seeking for opioid use disorder (OUD) therapy, your doctor may recommend Zubsolv (buprenorphine and naloxone). This prescription medication can ... read more...assist alleviate withdrawal symptoms. When you quit taking opioid medicines, you may experience similar symptoms. Zubsolv is a tablet that dissolves in the mouth. On the first day of therapy, you may take Zubsolv a couple times. After the first day, you'll most likely take Zubsolv once a day. Zubsolv is often used as a long-term therapy for OUD. So, here are some information that you should know about Zubsolv.
-
Zubsolv is a prescription medication intended to treat individuals who are addicted to opioid medications (prescription or illicit) as part of a comprehensive treatment program that includes counseling and behavioral therapy.
Zubsolv is classified as a controlled substance (CIII) because it includes buprenorphine, which is a drug of abuse for persons who abuse prescription medications or illicit narcotics.

opiates.com https://www.youtube.com/watch?v=8FABu6ubCJU -
Zubsolv is a brand (trade) name for a sublingual tablet containing buprenorphine and naloxone that may be used to treat opioid addiction. Buprenorphine is an opioid (narcotic) with a distinct and complicated mechanism of action that involves partial binding to mu opioid receptors and total binding to kappa opioid receptors. Opioid receptors have three major effects: they reduce breathing (respiratory depression), they provide euphoria (intense happiness or excitement), and they reduce pain. Because of the way buprenorphine binds, it can fulfill opioid cravings without generating euphoria or substantial respiratory depression. Furthermore, while it is attached to opioid receptors, other opioids (such as heroin and oxycodone) are unable to bind.
It also lingers on the receptors for a longer period of time - roughly three days - making it an excellent candidate for usage in opioid addiction treatment programs. To dissuade abuse, this combo pill contains Naloxone. If drug users attempt to convert the pill into an injectable version, they will experience opioid withdrawal since naloxone is a highly potent mu opioid receptor blocker. When administered sublingually, naloxone has almost no additional impact. Zubsolv is a medication of the family known as combined opioid/opioid antagonists. It is also known as a narcotic analgesic combination.

opioidtreatment.net 
opioidtreatment.net -
It has the potential to be utilized to treat opiate addiction (opioid misuse disorder). Zubsolv is part of a comprehensive treatment plan that also includes counseling and emotional assistance. A single daily dosage is administered. Treatment should begin under physician supervision and gradually move to unsupervised administration when the patient's clinical stability allows. The dosage of take-home medicine should take the patient's stability, as well as their home security and other circumstances, into account.
The dose can be gradually increased until the optimal amount is established that keeps the patient in therapy while suppressing opioid withdrawal symptoms. Instead of buprenorphine monotherapy, it may be utilized as first treatment in people addicted to heroin and other short-acting opioids. To avoid triggering opioid withdrawal syndrome, Zubsolv should be taken as soon as indications of withdrawal occur but no later than six hours after the last opioid usage.
Patients who are addicted on methadone or long-acting opioids may be more vulnerable to triggered and extended withdrawal following Zubsolv induction and should instead be started on buprenorphine monotherapy. After initiating buprenorphine monotherapy, the patient might be switched to once-daily Zubsolv. Induction and maintenance dose guidelines may be found in the product information.

drleeds.com 
drleeds.com -
If you are between the ages of 18 and 60, do not take any other medications, and have no other medical issues, you are more likely to have the following negative effects:
- The most common adverse effects recorded include headaches, gastrointestinal difficulties (e.g., constipation, nausea, or vomiting), pain, withdrawal syndrome, infection, sleeplessness, perspiration, and rhinitis. Elevations in liver enzymes (for example, alanine aminotransferase, aspartate aminotransferase, gamma-glutamyl transferase) and blood creatine phosphokinase have also been documented.
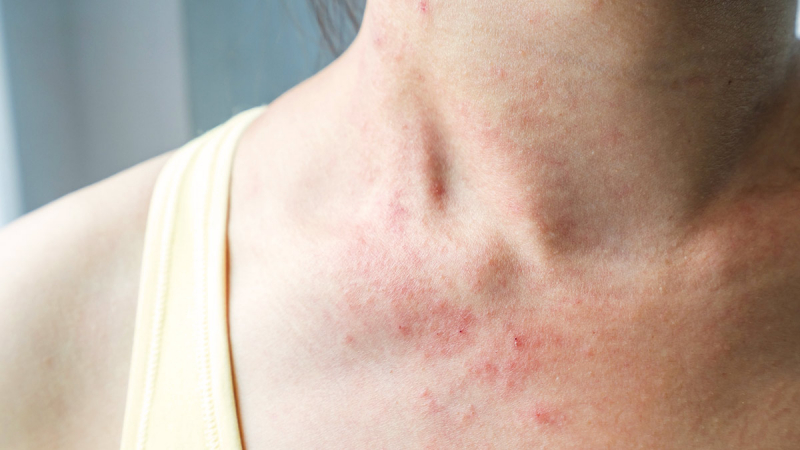
- Has the potential to cause significant, maybe fatal respiratory issues. This is especially common in those who abuse Zubsolv or when it is used with other drugs that impair respiration, such as benzodiazepines. Children, the elderly or disabled, and individuals with pre-existing respiratory difficulties are also more vulnerable.
- Even at the recommended dosage, it may become habit-forming. Zubsolv is a category III prohibited drug that must be prescribed and distributed with prudence to reduce the risk of overuse, abuse, or diversion.
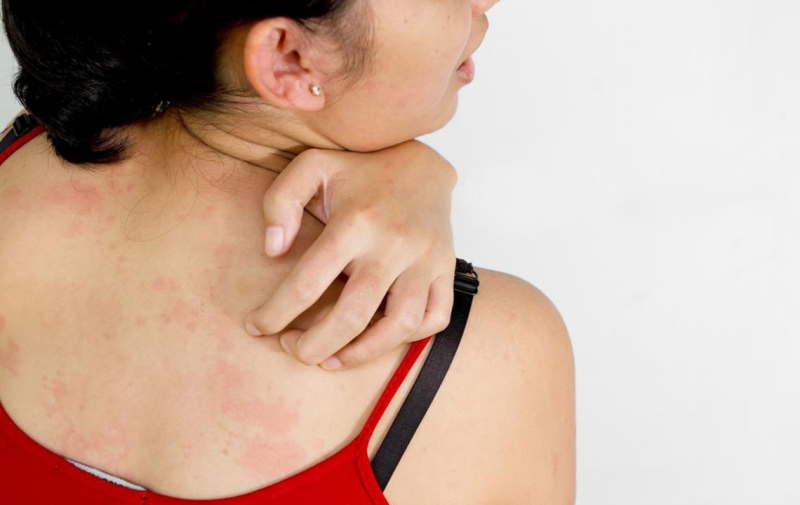
- There have been reports of allergic responses (including breathing difficulty, rash, and anaphylactic shock) and liver damage (hepatitis). Zubsolv should not be used in individuals who have a history of buprenorphine hypersensitivity.
It can make you sleepy or dizzy, and it can impair your ability to drive or operate machines. It is best to avoid alcohol.
May result in a dramatic drop in blood pressure or a quick reduction in blood pressure upon rising from a seated position. - At larger doses, there is a risk of cardiac rhythm abnormalities (such as QT prolongation).

SOTT.net 
holotropicbreathwork.net -
Take Zubsolv precisely as directed by your doctor. Your doctor may adjust your dose based on how you respond. Change your dosage only if your doctor instructs you to. Take Zubsolv no more frequently than your doctor has suggested. You may be given two or more Zubsolv sublingual pills at the same time.
Take Zubsolv once a day after induction (your first day of dosage). The pill should not be sliced, crushed, broken, chewed, or swallowed. Your doctor should show you how to properly take Zubsolv. Every time you take a Zubsolv dosage, follow the same instructions.
Zubsolv is available in a blister pack of ten blister units. Each blister pack includes a Zubsolv pill. Do not chew or swallow the pill while Zubsolv is dissolving since the drug will not function as well. Wait until the Zubsolv pill has completely dissolved before eating or drinking anything.
Talking as the tablet dissolves might impair the absorption of the medicine in Zubsolv. If you forget to take your Zubsolv, take it as soon as you recall. If your next dosage is approaching, omit the missed dose and take the following dose at your normal time. Unless your doctor instructs you otherwise, do not take two doses at the same time. Call your doctor if you are unsure about your dose.

humidcreative.com 
humidcreative.com -
Do not drive, operate heavy machinery, or engage in any other potentially hazardous activity until you have determined how this drug affects you. Drowsiness and sluggish response times can be caused by buprenorphine. This is most common in the first few weeks of therapy when your dose is altered, but it can also happen if you consume alcohol or use other sedative medicines while using Zubsolv.
You should not consume alcohol while taking Zubsolv since it might cause loss of consciousness or possibly death.
helpmegetoffdrugs.com https://www.youtube.com/watch?v=Za21dkYZ55c -
Medicines that interact with Zubsolv may reduce its impact, shorten its duration of action, enhance adverse effects, or have no effect when combined. An interaction between two drugs may not necessarily necessitate the discontinuation of one of them; nonetheless, it can. Consult your doctor about how to handle medication interactions.
Zubsolv may interact with the following medications:
- 5-hydroxytryptophan
- albuterol
- antibiotics, such as azithromycin, clarithromycin, erythromycin, and norfloxacin
- antidepressants, such as tricyclic antidepressants (eg, amitriptyline), monoamine oxidase inhibitors (eg, isocarboxazid, phenelzine, and tranylcypromine), or SSRIs (eg, fluoxetine, sertraline)
- antifungal agents, such as itraconazole and ketoconazole
- anticonvulsants, such as brivaracetam, carbamazepine, divalproex, lamotrigine, phenytoin, phenobarbital, or primidone
- antipsychotics (such as butyrophenones, phenothiazines, or thioxanthenes) and atypical antipsychotics (eg, aripiprazole, olanzapine, quetiapine, ziprasidone)
- any medication that may cause drowsiness, such as benzodiazepines (eg, alprazolam, diazepam, lorazepam), sleeping pills (such as zolpidem or ziprasidone), or first-generation antihistamines (such as doxylamine or promethazine)
humidcreative.com 
humidcreative.com